An electrode made for two Greek words one is called Elektron means electricity-driven, and hodos mean ways. An electrode is an electrical conductor, and it makes contact with nonmetallic parts of the circuit like electrolyte, semiconductor, and vacuum. Consider electrochemical cell; the electrode is used as one half cell electrode considered an anode that is left-hand side and another half cell considered a cathode that is the right-hand side. On this anode, the electron is lost, so an oxidation reaction takes place on the other side, called cathode electron gain, so a reduction reaction takes place. When both the oxidation and reduction process occurs simultaneously, it is called a redox reaction.
Table of Contents
ToggleMainly five types of the electrode
- Electrode reversible with respect to cation
- Electrode reversible with respect to anion
- Redox electrode
- Gas electrode
- Metal amalgam electrode
Electrode reversible with respect to cation
This type of electrode is also known as a Metal/Metal ion electrode. When a metal strip (electrode) is kept in a solution, it consists have its own cation. For example, a Zinc metal strip is kept in a Zinc sulphate solution, or a Copper strip is put in a Copper sulphate solution. It is known as the Daniel cell or Electrochemical cell Example:
| Metal is univalent electrode reaction represent as, M(s)/M+n(aq) When metal is bivalent electrode reaction represent as, Zn(s) /Zn+2(aq) Oxidation: Zn(s) – 2e-↔ Zn+2(aq) E0oxidation = 0.76 volts Reduction: Zn+2(aq)+ 2e-↔ Zn(s) E0 reduction = -0.76 volts Oxidation : Cu(s) – 2-e↔ Cu+2(aq) E0 oxidation = 0.34 volts Reduction : Cu+2(aq) +2e- ↔ Cu(s) E0 reduction = -0.34 volts |
Electrode reversible with respect to anion
This electrode is known as a metal/metal sparingly soluble salt/salt anion electrode. This electrode type is formed when the metal rod is kept in contact with its sparingly soluble salt and common anion-containing solution. Example:
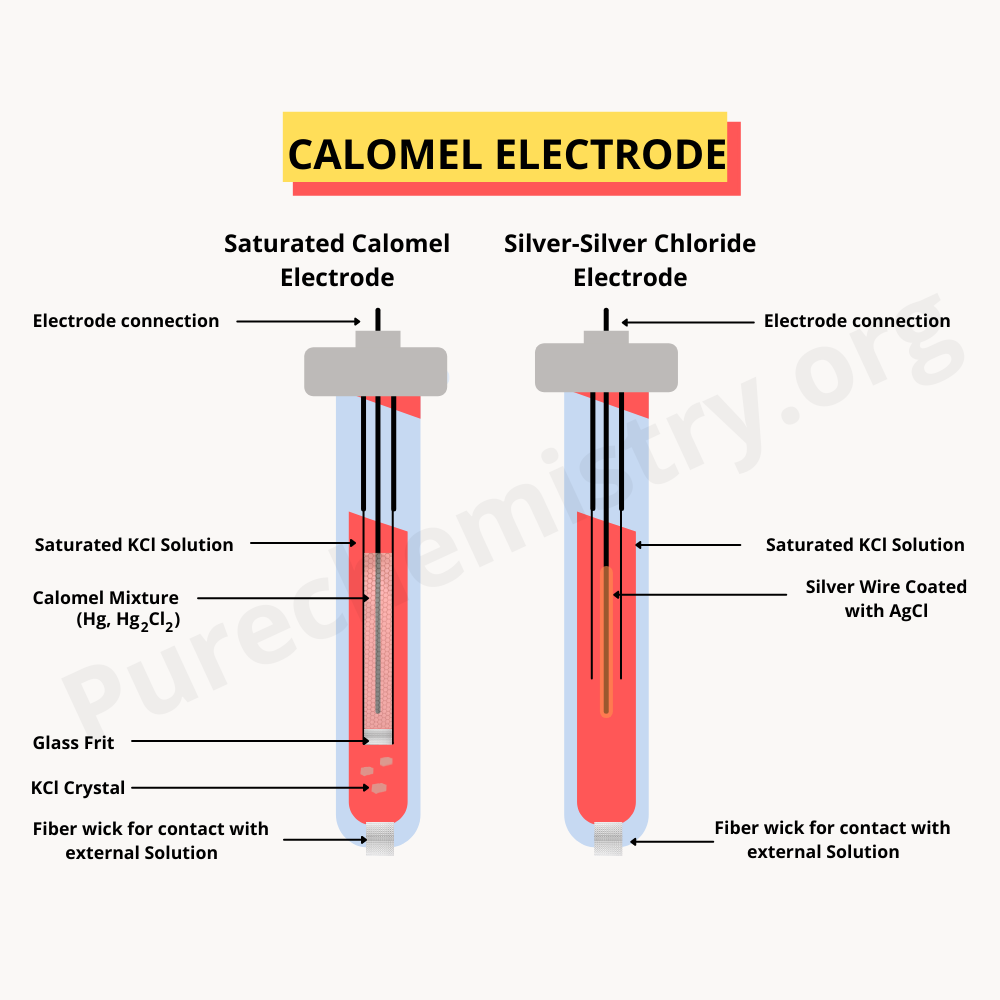
| Silver wire is coated with a paste of Agcl then kept in a solution that consists of Cl– ions. Cell represents as, Ag(s) /AgCl(s) / Cl–(aq) This type of electrode is the best example is the calomel electrode |
The calomel electrode is used as a reference electrode. It is metal amalgam electrode depends on the concentration of KCl solution. Construction of calomel electrode is very simple by filling at the end of glass tube pure Hg than the paste of Hg/Hg2Cl2 on it fills in the whole tube with a saturated solution of KCl. One glass capillary is kept in a glass tube consisting of a platinum wire and is in contact with pure Hg at the end. The cell is denoted as, Pt/Hg/Hg & Hg2Cl2 paste/Sat.KCl On the one side of electrode reduction reaction occurs, Pure Hg(s) and sparingly soluble mercurous chloride discharged at the electrode, increasing the chloride ion concentration in solution. This reaction is denoted as, Hg+2(aq) + 2e- ↔ 2Hg(l) Hg2Cl2(S) ↔ Hg+2(aq) + 2Cl– (aq)
On the other side, an Oxidation reaction occurs formed two moles of Hg+2 ions and this ion reacts with Cl– to formed Hg2Cl2. For this reaction, decrease the concentration of chloride ions in the solution. 2 Hg(l) ↔ Hg+2(aq) + 2e- Hg+2(aq) + 2Cl– (aq) ↔ Hg2Cl2(S) Overall, Calomel electrode reaction denoted as, Hg2Cl2(S) + 2e- ↔ 2 Hg(l) + 2Cl– (l) This electrode is known as reversible with respect to anion (chloride ion).
Redox Electrode (Oxidation – Reduction Electrode)
Redox electrode is also known as an inert electrode. The setup electrode is a metal strip kept in a solution consisting of its own metal ion having different valence due to this process potential develops. Example:
|
Sn/ Sn+2 , Sn+4 Fe/Fe+2 , Fe+3 The reaction did in electrode on anode oxidation reaction, and cathode reduction reaction (Redox reaction) occurs; Oxidation : Sn+2 – 2e– ↔ Sn+4(aq) Reduction : Sn+4(aq) + 2e– ↔ Sn+2 (aq) The type of redox electrode known as Quinhydrone electrode is Another reference electrode but slightly different. |
This reference electrode is an equimolar mixture of quinon and hydroxy quinon. Which are organic compounds. The reaction between two compounds is,

|
To measure the concentration of [H+] in the solution, add quinhydrone powder to make a saturated solution and keep the platinum electrode in this solution. Another beaker filled with saturated KCl solution kept the calomel electrode. Both solutions connect via a salt bridge. Example:
|
Pt/Hg&Hg2Cl2paste/Saturated KCl/[H+] solution saturated with quinhydrone/pt. This electrode is used in Potentiometric titration. The advantage of this electrode is easily prepared, and the disadvantage of this electrode is used only for those whose solution is less than 8.0 pH above 8.0 pH. This electrode does not work. |

Gas electrode
The gas electrode is known as a non-metal electrode. In this electrode, pure H2 gas passed over pt foil is kept in [H + ] solution then this type of electrode is used. There are three types of gas electrodes.
Hydrogen Gas electrode
This electrode is used as a Primary reference electrode to determine unknown cell potential. Another name of this electrode is known as standard hydrogen electrode (SHE). This electrode can be prepared using a glass tube with two small whole at the bottom another hole upper side; a glass capillary is passed from this tube and consists of a platinum wire, and at the bottom side, platinum foil coated with platinum black. The whole part is kept in a solution having a 1M concentration of hydrogen ion. The top of the tube continuously passed 1 atmospheric pressure hydrogen gas, which bubbled out in the solution. The cell can be represented as, Pt/H2(g) 1atm/ H+ (aq)(1M)// // symbol represent Salt bridge A reaction occurs in this cell; Oxidation: ½ H2(g) – e– ↔ ½ H2(aq) Reduction: ½ H2(aq) + e– ↔ ½ H2(g)
In this electrode, oxidation and reduction reaction takes place on platinum foil surface. SHE(standard hydrogen electrode) has a fixed potential of 0.000 volts at 25°C. The advantage of this electrode is used to prepare the emf(electromotive force) series. One drawback of the electrode is not easy to continues passed hydrogen gas at 1 atmospheric pressure.

Chlorine Electrode:
Chlorine electrode work as same for hydrogen gas electrode instead of hydrogen in this electrode using chlorine gas. At 1 atmospheric pressure, chlorine gas passed into a tube which kept in hydrochloride solution. Chlorine gas electrode represents as; Pt/Cl2(g) /Cl-2 (aq) // A reaction occurs in this cell: Oxidation: ½ Cl2 (g) – e– ↔ ½ Cl+2 (aq) Reduction: ½ Cl+2(aq) + e– ↔ ½ Cl2(g) This electrode is reversible in the presence of anion (chloride ion).
Oxygen Electrode
Oxygen electrodes also work as both electrodes like hydrogen and chloride electrode, but in this electrode, oxygen gas is passed at a particular pressure in the glass capillary. Tube put in a solution containing hydroxyl ions. Oxygen gas electrode represents as ; Pt/O2(g) / OH–(aq) // A reaction occurs in this cell : Oxidation: 4OH–(aq) ↔ 2H2O + O2(g)+ 4e– Reduction: 2H2O + O2(g) + 4e– ↔ 4OH– (aq) This electrode does not behave as a truly reversible electrode because equilibrium is not easily established between oxygen gas and hydroxyl ions in the solution.
Metal amalgam electrode
This type of electrode-formed metal strip is kept in metal amalgam solution. Zn/Zn in Hg or Cd/Cd in Hg

Citation & Reference:
|
Citation: (Sheladiya, 2021) Reference this article as (APA): Sheladiya, B. S. (2021, August 30). TYPES OF REVERSIBLE ELECTRODES. Retrieved from www.purechemistry.org: https://www.purechemistry.org/types-of-reversible-electrodes/ |